[vc_row][vc_column][vc_custom_heading text=”What is Alachlor?”][vc_column_text]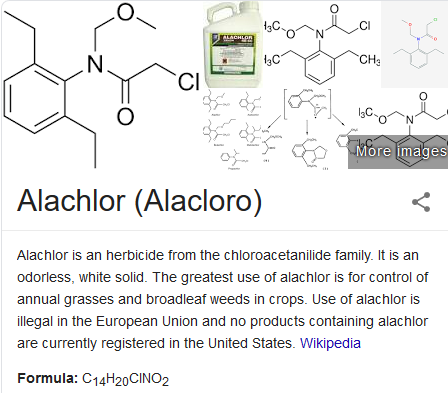 Alachlor is an aniline herbicide from the chloroacetanilide family. It is an odorless, white solid. It is an aromatic amide that is N-(2,6-diethylphenyl)acetamide substituted by a methoxymethyl group at the nitrogen atom while one of the hydrogens of the methyl group has been replaced by a chlorine atom. It has a role as a herbicide, an environmental contaminant and a xenobiotic. It is an organochlorine compound, a monocarboxylic acid amide, and an aromatic amide. It derives from a N-phenylacetamide. It is used to control annual grasses and certain broadleaf weeds in field corn, soybeans, and peanuts. It is a selective systemic herbicide, absorbed by germinating shoots and by roots, the compound works by interfering with a plant’s ability to synthesize protein and by interfering with root elongation. The compound is found in a variety of commercial herbicides including Lasso, Lariat, and Crop Star. It also mixes well with other herbicides such as Bullet, Freedom, and Rasta and in mixed formulations with atrazine, glyphosate, trifluralin and imazaquin. It is a selective, systemic herbicide, absorbed by germinating shoots and by roots. It’s chemical formula is C14H20ClNO2 and it can be known as 2-chloro-N-(2,6-diethylphenyl)-N-(methoxymethyl)acetamide / alachlor, N-methoxymethyl-2′,6′-diethyl-2-chloroacetanilide and 15972-60-8; butachlor, N-(butoxyethyl)-2-chloro-2′,6′-diethylacetanilide and 23184-66-99.[/vc_column_text][vc_custom_heading text=”What is acetochlor used for?”][vc_column_text]Alachlor is used pre- or early post-emergence to control annual grasses and many broadleaved weeds mainly in maize, but also cotton, brassicas, oilseed rape, peanuts, radish, soybeans, and sugar-cane.
Alachlor is an aniline herbicide from the chloroacetanilide family. It is an odorless, white solid. It is an aromatic amide that is N-(2,6-diethylphenyl)acetamide substituted by a methoxymethyl group at the nitrogen atom while one of the hydrogens of the methyl group has been replaced by a chlorine atom. It has a role as a herbicide, an environmental contaminant and a xenobiotic. It is an organochlorine compound, a monocarboxylic acid amide, and an aromatic amide. It derives from a N-phenylacetamide. It is used to control annual grasses and certain broadleaf weeds in field corn, soybeans, and peanuts. It is a selective systemic herbicide, absorbed by germinating shoots and by roots, the compound works by interfering with a plant’s ability to synthesize protein and by interfering with root elongation. The compound is found in a variety of commercial herbicides including Lasso, Lariat, and Crop Star. It also mixes well with other herbicides such as Bullet, Freedom, and Rasta and in mixed formulations with atrazine, glyphosate, trifluralin and imazaquin. It is a selective, systemic herbicide, absorbed by germinating shoots and by roots. It’s chemical formula is C14H20ClNO2 and it can be known as 2-chloro-N-(2,6-diethylphenyl)-N-(methoxymethyl)acetamide / alachlor, N-methoxymethyl-2′,6′-diethyl-2-chloroacetanilide and 15972-60-8; butachlor, N-(butoxyethyl)-2-chloro-2′,6′-diethylacetanilide and 23184-66-99.[/vc_column_text][vc_custom_heading text=”What is acetochlor used for?”][vc_column_text]Alachlor is used pre- or early post-emergence to control annual grasses and many broadleaved weeds mainly in maize, but also cotton, brassicas, oilseed rape, peanuts, radish, soybeans, and sugar-cane.
Alachlor was detected in rural domestic well water by EPA’s National Survey of Pesticides in Drinking Water Wells.
EPA’s Pesticides in Ground Water Database reports detections of alachlor in groundwater at concentrations above the MCL in at least 15 U.S. states.
Alachlor is also often used in high school chemistry classrooms as a reactant in demonstrations such as the combustion of magnesium.
Alachlor can be used as a substitution for methane gas in such an experiment when gas is not available.[/vc_column_text][vc_custom_heading text=”How do Alachlor works?”][vc_column_text]Its mode of action is elongase inhibition, and inhibition of geranylgeranyl pyrophosphate (GGPP) cyclization enzymes, part of the gibberellin pathway.
Stated more simply, it works by interfering with a plant’s ability to produce protein and by interfering with root growth.
The use of alachlor is illegal in the European Union and no products containing alachlor are currently registered in the United States.[/vc_column_text][vc_custom_heading text=”Is Alachlor harmful?”][vc_column_text]Alachlor is a Restricted Use Pesticides may be purchased and used only by certified applicators.
It carries the signal word DANGER on the label, not because of its acute oral toxicity, which is fairly low, but rather due to its potential to cause cancer in laboratory animals.
The United States Environmental Protection Agency (EPA) classifies the herbicide as toxicity class III – slightly toxic.
The Maximum Contaminant Level Goal (MCLG) for Alachlor is zero, to prevent long-term effects, and the Maximum Contaminant Level (MCL) for drinking water is two parts per billion (2 ppb).
The EPA cited the following long-term effects for exposures at levels above the MCL in drinking water exposed to runoff from herbicide used on row crops: slight skin and eye irritation; at lifetime exposure to levels above the MCL: potential damage to liver, kidney, spleen; lining of nose and eyelids; cancer.
In Australian law and it is listed as a Schedule 7 (Dangerous Poison) substance, so the access, use, and storage are strictly controlled under state and territory law.
Since 2006, the use of alachlor as a herbicide has been banned also in the European Union.
When we are talking about its harmful potential, other than cancer, liver toxicity, and eye lesions are the principal chronic toxic effects to organs seen in various studies.[/vc_column_text][vc_custom_heading text=”Alachlor in the environment”][vc_column_text]Alachlor persists in soil from two weeks to a month, depending on soil type and climate. About half of the compound remains in the soil after eight days.
The main means of degradation is by soil microbes, It has moderate mobility in sandy and silty soils and thus can migrate to groundwater.
The single largest groundwater testing program (National Alachlor Well Water Survey) for a pesticide was conducted for alachlor throughout the last half of the 1980s. Over six million private and domestic wells were tested for the presence of the compound and less than one percent of all of the wells had detectable levels of alachlor.
Detection of the chemical in well water was more common where the herbicide was used more intensively.
No parent compound was found after three weeks when alachlor was applied to plants.
It was rapidly metabolized to water-soluble products with the acid product accumulating in the plant.
Alachlor should not pose a problem for bees when used as directed. The product is practically non-toxic to earthworms.
Food does not appear to be a major route of exposure for the general population since
residues of alachlor in food are usually below the detection limit.[/vc_column_text][vc_video link=”https://www.youtube.com/watch?v=ACxe6IVtPIs”][/vc_column][/vc_row]